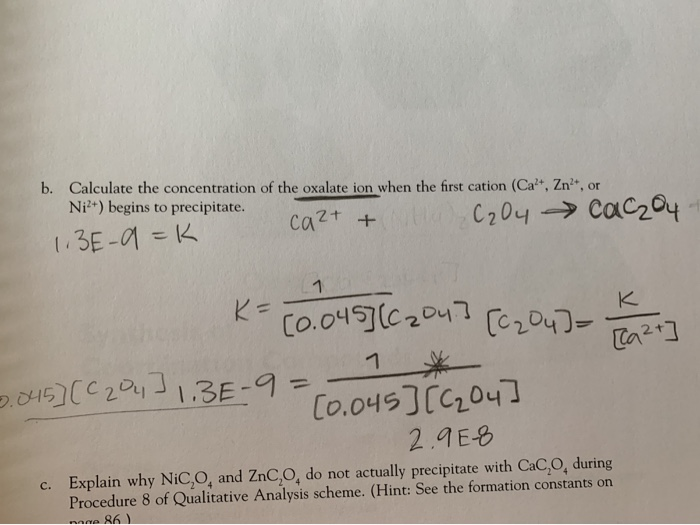Calculate the maximum normality of a solution containing 13.4 g of sodium oxalate in 100ml solution.

Calculate the normality of a solution containing 13.4 g fo sodium oxalate in 100mL solution. - YouTube

Chlorthalidone with potassium citrate decreases calcium oxalate stones and increases bone quality in genetic hypercalciuric stone-forming rats - ScienceDirect

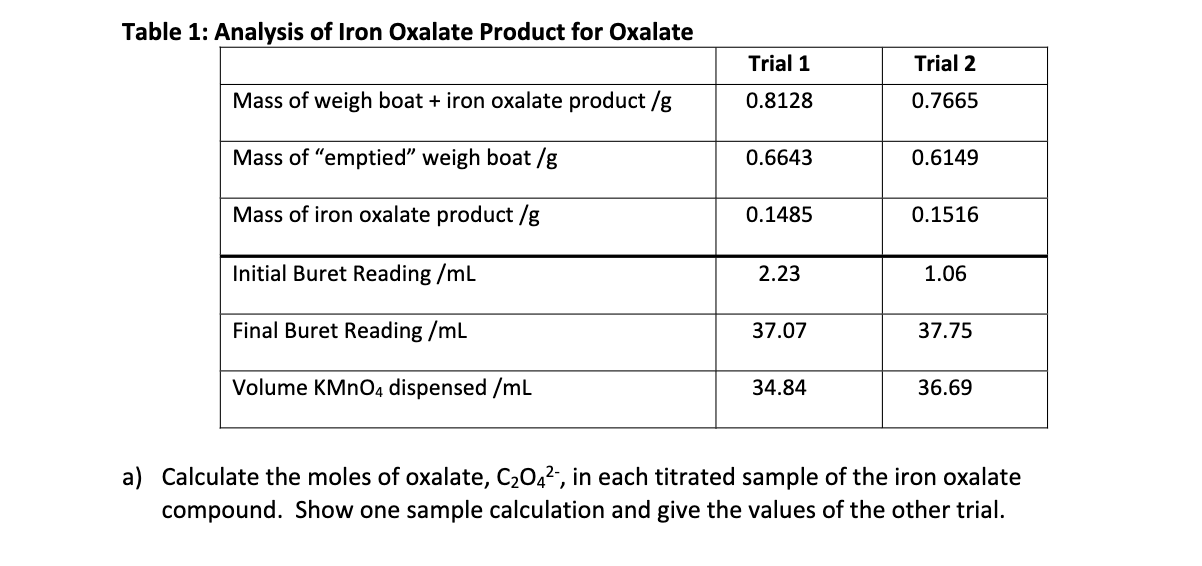
SOLVED: The composition of a hydrated potassium-salt of a cobalt(IIl)- oxalate complex ion conforms to the general formula: KCo(CO4)3wHO A 0.4925-g sample of this compound required 46.52 mL of 0.02667M KMn04 solution for

0.5 g of an oxalate was dissolved in water and the solution made to 100 mL. On titration 10 mL of this solution required 15 mL of (N)/(20)KMnO(4). Calculate the percentage of

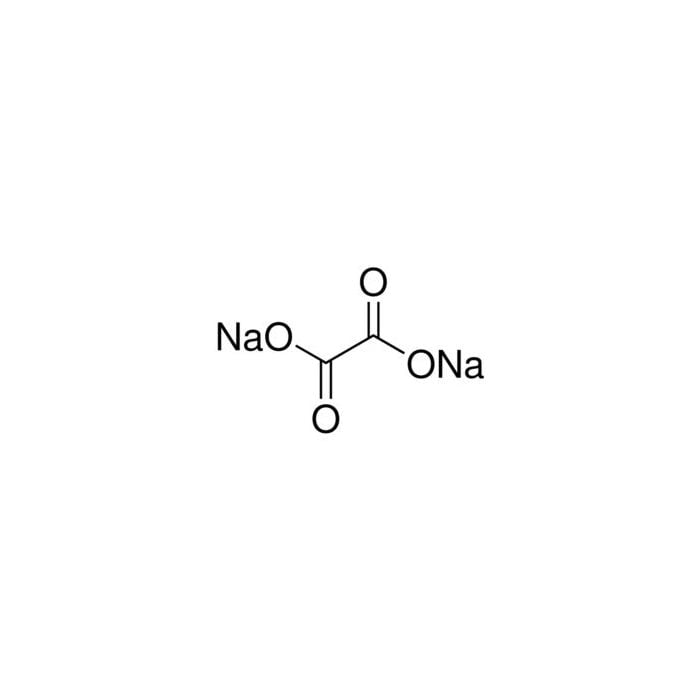



![Oxalate [C2O4^(-2)] - Structure, Molecular Mass, Properties & Uses Oxalate [C2O4^(-2)] - Structure, Molecular Mass, Properties & Uses](https://cdn1.byjus.com/wp-content/uploads/2019/04/oxalate-structure.png)


![Silver Oxalate [Ag2C2O4] Molecular Weight Calculation - Laboratory Notes Silver Oxalate [Ag2C2O4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/silver-oxalate-molecular-weight-calculation-300x189.jpg)

![ANSWERED] A chemist prepares a solution of zinc oxa... - Physical Chemistry ANSWERED] A chemist prepares a solution of zinc oxa... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/72593655-1657405017.6312482.jpeg)